SAP BRH for Life Sciences
OUR SOLUTUIONS
SAP Batch Release Hub for Life Sciences
What is SAP Batch Release Hub?
The life sciences industry is highly regulated and must follow strict policies, especially around quality control. The batch release process is essential for maintaining this quality. Before active pharmaceutical ingredients (APIs) are used in manufacturing, they must pass several checks before the final drug product can be released to the market. Because of these requirements, manufacturers need a digital tool that helps them meet all regulations while reducing the time it takes to bring products to market.
SAP Batch Release Hub for Life Sciences, supported by Saptix, is an advanced digital solution that automatically gathers and reviews all the quality and regulatory data needed for batch release. Its user-friendly interface helps quality control teams make faster and more accurate batch release decisions.
This hub reduces risks for the entire team, helps lower inventory costs, and speeds up market delivery with more consistent order fulfillment and optimized batch sizes. As a result, it boosts company productivity, supports sustainable logistics, and improves financial performance.
Batch Release Process Challenges
Low-Quality Product Recalls
Manufacturers are responsible for any harm caused by defective products. Poor quality can lead to serious health risks, high costs, and damage to the company’s reputation.
Lack of Process Visibility
The batch release process needs full visibility to spot and address risks before products are shipped. Good tracking is essential for manufacturers to maintain safety and reduce compliance issues.
Unintentional Human Errors
Mistakes like incorrect assembly, counting errors, wrong labeling, or subjective decisions can cause injuries or property damage. Reducing human errors is a top priority for manufacturers.
Legacy Siloed Systems
Old, disconnected systems create problems such as duplicate data, poor communication, access risks, and other process challenges. This leads to longer lead times and makes it hard to see the overall business picture.
Follow Stringent Quality Control with SAP Batch Release Hub
Complete View of the Batch Release Process
Improved Compliance with Less Manual Work
Transparent Release Check Results
Adopt a Go-To Framework for Supreme Product Liability
Depending on your business needs, SAP Batch Release Hub for Life Sciences, supported by Saptix, can be integrated with a wide range of source systems, including SAP and non-SAP applications, using public APIs and flexible integration options
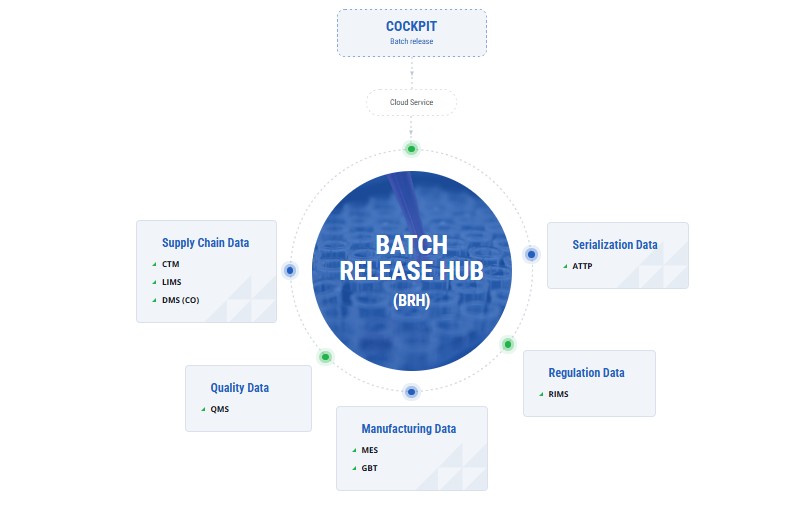
How Can You Benefit From Using SAP Batch Release Hub?
20% Reduction in Quality Costs – $20 Million Savings
The efficiency improvements achieved through QM reduce the time spent checking batches with exceptions. This leads to lower operating costs and, in turn, lowers the overall cost of quality. For example, a 20% reduction in quality costs can generate an average savings of around $20 million for a company.
How We Can Help
We offer end-to-end services for SAP BRH:
Implementation Roadmap
We follow the SAP Activate methodology, which divides the implementation of SAP Batch Release Hub for Life Sciences into six key stages:
FAQ
-
Can SAP BRH handle multistep batch release processes?
Yes. SAP BRH supports complex workflows with multistep approvals and ensures compliance with strict release protocols.
-
How does SAP BRH support quality management?
SAP BRH integrates with quality management systems (QMS), allowing businesses to monitor quality, maintain consistency, and ensure compliance across all batches.
-
How does SAP BRH integrate with other SAP solutions?
SAP BRH connects with SAP ERP, SAP QMS, and other life sciences solutions, creating a unified platform for efficient batch and quality management.
-
Can SAP BRH provide batch traceability?
Yes. SAP BRH delivers complete batch traceability, enabling companies to manage recalls effectively and meet regulatory requirements.
-
Does SAP BRH support electronic batch record (EBR) management?
Yes. SAP BRH offers electronic batch record management, reducing paperwork, and improving data accuracy, accessibility, and compliance.
-
Can SAP BRH be integrated with existing QMS and ERP solutions?
Yes. SAP BRH integrates seamlessly with existing quality management and ERP systems. This ensures consistent data exchange, better workflow alignment, and improved operational efficiency across batch release, quality, and manufacturing processes.
-
How does SAP BRH improve collaboration between quality assurance, production, and regulatory teams?
SAP BRH provides a single platform for QA, production, and regulatory affairs teams. It enables real-time collaboration, electronic signatures, and review of batch records, helping speed up batch release cycles while maintaining compliance.